3 Ways to Determine Order of Reaction
Introduction:
In the world of chemistry, the study of reaction rates and mechanisms is of utmost importance. It helps us understand how chemical transformations occur and allows us to control processes for industrial and scientific purposes. One of the fundamental aspects of studying reaction kinetics is determining the order of a reaction. The order of a reaction is the sum of the exponents in the rate law and refers to how changes in concentrations impact the overall rate of the reaction. In this article, we will discuss three methods to determine the order of a reaction: graphical representation, initial rates method, and half-life method.
1.Graphical Representation:
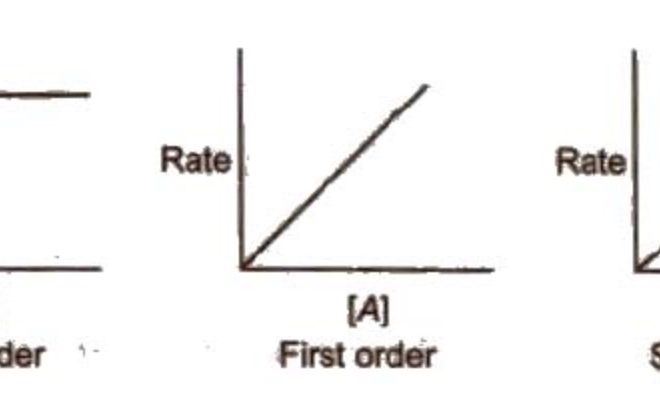
A popular method to deduce a reaction’s order is by plotting graphs of concentration versus time for specific reactants. By observing the trends in these graphs, one can deduce if a reaction exhibits zero, first, or second-order kinetics.
– Zero-order reactions: When plotted as concentration vs time, a zero-order reaction displays a straight line with a negative slope. This indicates that the rate remains constant throughout the process.
– First-order reactions: A first-order reaction shows an exponential decay when plotted as concentration vs time. When plotted as ln(concentration) vs. time, it forms a straight line with a negative slope.
– Second-order reactions: For second-order reactions, when reciprocal concentration (1/concentration) is plotted against time, it results in a straight line with a positive slope.
2.Initial Rates Method:
The initial rates method requires having multiple sets of experimental data where different concentrations are used under identical experimental conditions. The purpose is to observe how changes in concentrations affect the initial rates (the rate measured at t = 0). A change in concentration will result in changes to the initial rate which corresponds to an exponent in the rate law.
To use this method:
– Calculate individual initial rates for each experiment set.
– Compare any two sets of experimental data to deduce the exponent of a reactant in the rate law.
– Iterate this process for all reactants, and sum up the exponents to determine the overall order of the reaction.
3.Half-Life Method:
Half-life (t1/2) is the time taken for the concentration of a reactant to be reduced to half its initial value. The half-life method is particularly useful when working with first-order reactions.
– For a first-order reaction, the half-life remains constant irrespective of the concentration.
– If a reaction’s half-life does not change when its concentration is varied, it is likely that it follows first-order kinetics.
– In contrast, zero and second-order reactions exhibit varying half-lives based on their respective concentrations. Thus, if a reaction doesn’t exhibit a constant half-life, alternative methods are necessary to determine its order.
Conclusion:
Determining the order of a reaction is an essential step in understanding and describing chemical kinetics. By using graphical representation, initial rates method, and half-life method, chemists can classify reactions into standard orders and build accurate rate laws to predict and control their behavior. Each method has its own advantages and limitations; therefore, employing multiple methods helps confirm results and reach accurate conclusions in determining a reaction’s order.






