How to calculate neutrons in an atom
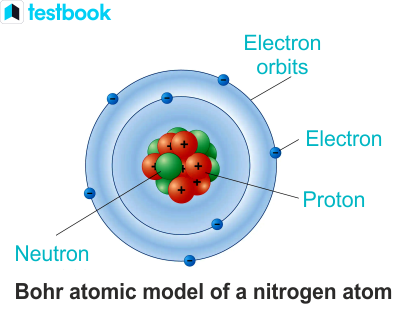
In the study of chemistry and physics, understanding the structure of atoms is essential for gaining insights into their properties and behavior. One crucial component of an atom is the neutron – a subatomic particle carrying no charge. Neutrons play a vital role in determining the atomic mass and stability of an atom. This article will guide you through the process of calculating the number of neutrons in an atom.
To calculate neutrons in an atom, you need two key pieces of information – atomic number (Z) and mass number (A).
Here’s a step-by-step guide to help you with this process:
1. Identify Atomic Number (Z):
The atomic number represents the number of protons in an element’s nucleus. It also defines its chemical properties and placement on the periodic table. To identify the atomic number, refer to the periodic table, where each element has its unique atomic number listed above or below its symbol.
Example: If you want to calculate the neutrons in a carbon atom, find carbon on the periodic table, which has an atomic number (Z) of 6.
2. Determine Mass Number (A):
The mass number is defined as the total number of protons and neutrons in an atom’s nucleus. For a given isotope or stable element, you can often find this information on periodic tables or online resources by searching for its name, followed by “isotope” or “mass numbers”. Alternatively, it could be provided explicitly; perhaps as part of a question.
Example: For our carbon example, we’ll consider Carbon-12 (one of its isotopes), which has a mass number (A) of 12.
3. Calculate Neutrons:
Finally, you can compute the total neutrons in an atom by subtracting its atomic number (Z) from its mass number (A)
Formula: Neutrons (N) = Mass Number (A) – Atomic Number (Z)
Example: For Carbon-12,
Neutrons (N) = Mass Number (A) – Atomic Number (Z)
Neutrons (N) = 12 (A) – 6 (Z)
Neutrons (N)= 6
In conclusion, in a Carbon-12 atom, there are 6 neutrons.
Now that you have a comprehensive understanding of how to calculate the neutrons in an atom, you can apply this knowledge in various scientific disciplines, such as chemistry, physics, and materials science. Furthermore, mastering the neutron calculation process will enable you to comprehend atomic structure, isotopes, and other fundamental concepts related to atoms. Happy calculating!






