How to calculate kcat
Introduction
The enzyme-catalyzed reaction rate is an essential parameter in molecular biology, biochemistry, and biotechnology. One of the main parameters used to describe enzyme efficiency is kcat, also known as the turnover number. It represents the number of substrate molecules that an enzyme can convert into products per unit time. This article will provide a step-by-step guide on how to calculate kcat.
Step 1: Understand the Michaelis-Menten Equation
To calculate kcat, you first need to familiarize yourself with the Michaelis-Menten equation:
v = (Vmax*[S]) / (Km + [S])
where v is the initial reaction velocity, Vmax is the maximum reaction velocity, [S] is the substrate concentration, and Km is the Michaelis constant.
Step 2: Determine Vmax
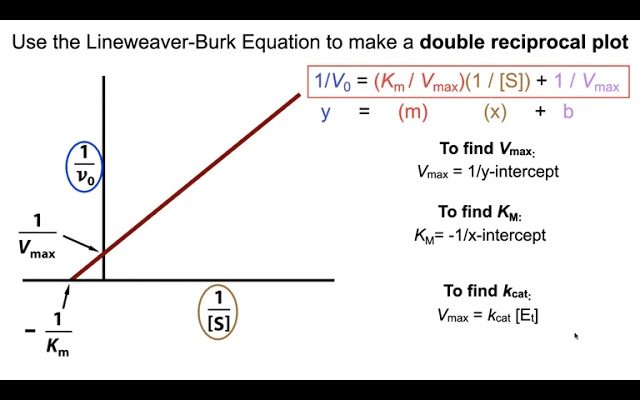
Vmax represents the maximum rate at which an enzyme can catalyze a reaction. You can determine Vmax from experimental data by conducting an enzyme kinetic assay at various substrate concentrations and plotting a graph of reaction velocity (v) versus substrate concentration ([S]). The value of v will approach Vmax as [S] increases. Vmax can be obtained by fitting a curve to these data points using non-linear regression analysis or using a graphical approach such as Lineweaver-Burk or Eadie-Hofstee plots.
Step 3: Calculate Enzyme Concentration
To calculate kcat, you first need to determine the concentration of active enzyme present in your experiment ([E]). This value can be determined through multiple methods, such as measuring protein absorbance at 280 nm or using a Bradford assay. Make sure that your enzyme is in its active form for accurate results.
Step 4: Calculate kcat
Finally, with both Vmax and [E] known, you can calculate kcat using the following equation:
kcat = Vmax / [E]
This will give you the turnover number for the enzyme, representing how efficient the enzyme is at converting substrate molecules into products over a given time.
Conclusion
Calculating kcat is crucial in understanding an enzyme’s efficiency and functionality. By following these steps, you can accurately determine kcat and use this information to compare different enzymes or evaluate the effect of various conditions on enzymatic activity. Knowledge of kcat can also aid in predicting reaction rates under different circumstances, designing improved enzymes, and guiding protein engineering efforts.